 Many industries have been using Lithium for different purposes, from medical services to, even, armed forces in several countries. It is a chemical element which is also known as Li on the periodic table. 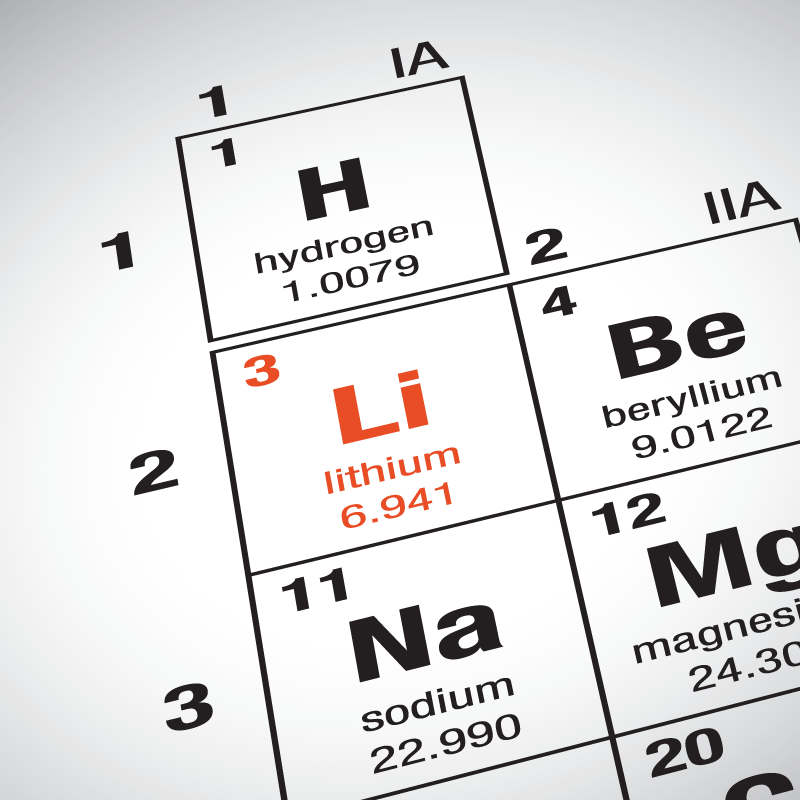
Under standard conditions, it is the least. Lithium is an alkali metal with anomic number 3. It’s moderately abundant and present in the Earth’s crust in 65 ppm (parts per million). It is a soft, silvery-white alkali metal. Lithium is a chemical element that occurs first in the alkalis of the periodic table. In the periodic table, it is located in group 1, among the. Refer to below table for Lithium Physical Properties DensityĠ.535 g/cm3(when liquid at m.p density is $0. Lithium is a chemical element with the symbol Li and atomic number 3. Lithium is a chemical element in the periodic table that has the symbol Li and atomic number 3. All possible symmetric arrangements of particles in three-dimensional space are described by the 230 space groups (219 distinct types, or 230 if chiral copies are considered distinct. Manic symptoms include hyperactivity, rushed speech, poor judgment, reduced need for sleep, aggression, and anger. Lithium a mood stabilizer that is a used to treat or control the manic episodes of bipolar disorder (manic depression ). The symmetry properties of the crystal are described by the concept of space groups. Lithium affects the flow of sodium through nerve and muscle cells in the body. It is the lightest metal and the lightest solid element. It is a soft, silvery-white alkali metal whose name comes from the Greek word lithos (which means stone). The positions of the atoms inside the unit cell are described by the set of atomic positions ( x i, y i, z i) measured from a reference lattice point. Lithium (Li) is a chemical element of the periodic table, located in group 1 and period 2, having the atomic number 3. Naturally occurring lithium (3 Li) is composed of two stable isotopes, lithium-6 and lithium-7, with the latter being far more abundant on Earth.Both of the natural isotopes have an unexpectedly low nuclear binding energy per nucleon (5 332.3312(3) MeV for lithium-6 and 5 606.4401(6) MeV for lithium-7) when compared with the adjacent lighter and heavier elements, helium (7 073.9156(4) MeV for. The unit cell is represented in terms of its lattice parameters, which are the lengths of the cell edges Lattice Constants (a, b and c) aĪnd the angles between them Lattice Angles (alpha, beta and gamma). The unit Cells repeats itself in three dimensional space to form the structure. The Crystal structure can be described in terms of its unit Cell. The solid state structure of Lithium is Body Centered Cubic. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
